Non-ferrous metal inhibitor
New, improved lubricants arrive on the market at regular intervals, and the formulation of these high-tech products, in most cases based on synthetic base oils and additives, also has direct consequences for lubricant analysis. That‘s why OELCHECK continually invests in innovative testing procedures and develops new analysis methods based primarily on test procedures that have become standard. A brand-new development is to inspect the remaining non-ferrous metal inhibitor content in waste oils using high-performance liquid chromatography (HPLC). Previously used by OELCHECK for coolant analysis, the HPLC method can now be applied for early detection of the breakdown of non-ferrous metal inhibitors used in gear oils to prevent non-ferrous metal wear.
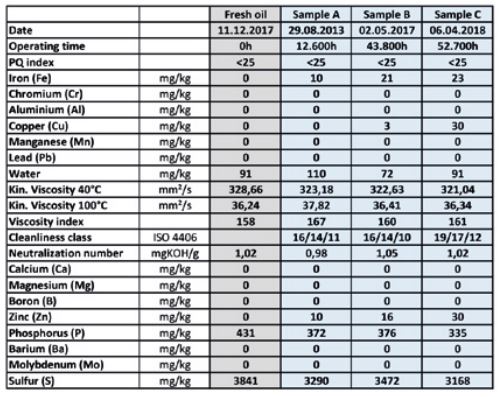
Our new analysis method of non-ferrous metal inhibitor (NFMI) content developed from an exciting case that initially had OELCHECK tribologists scratching their heads. Oil from the mean gear of a wind turbine filled with 600 litres of synthetic gear oil was inspected three times over a period of six years. The analysis values obtained showed that no oil change was necessary, as all of them were normal. However, a closer look at a sample of the oil that had been in use the longest revealed changes that were completely outside what was expected. The oil samples were taken at irregular intervals of 12,600, 43,800 and 52,700 operating hours, respectively. They revealed a change in the colour of the oil that was documented photographically in the lab report. Although there is no standardised evaluation for oil colour, this enables specialists to make an initial appraisal of oil changes in comparison with a fresh or previous sample. In this case, the first sample was still light and clear, but after that the oil had discoloured to a dark brown within the relatively short space of time up to 52,700 operating hours. As the remaining analysis values (with the exception of copper content) showed nothing unusual, the darkened oil alone revealed no clues to suggest that an oil change was needed. Nevertheless, the tribologists at OELCHECK were alarmed. Due in part to the copper content, they first examined the visual change since the last sample more closely under the microscope and compared it with the previous samples and with fresh oil.
In spite of this, neither the values obtained nor the FT-IR spectroscopy indicated a significant change in the oil or oil mixing. The viscosity and viscosity index had remained stable, the Neutralisation Number had barely risen, and the oil purity was within tolerable limits. Phosphorous and sulphur content – a typical part of the additive component – had only fallen slightly in the darkened sample. With the exception of copper, the wear metals showed no signs of significantly increased wear. Although the high copper value quite clearly indicated corrosive wear of components containing copper, such as roller bearing cages, copper alone is not a cause of oil discolouration.

Oil ageing and its traditional parameters
When inspecting wear values, OELCHECK uses an internal database where permissible boundary and warning values are defined for every system type on the basis of several hundreds of thousands of analysed samples. A comparison against the limit for copper levels resulted in the gear condition being assessed as „critical“; however, no clear cause for the unusually elevated copper content could be found, even with the more than 30 analysed values available. OELCHECK‘s tribologists consulted with lab management to find a method that could provide clarification. It was striking that both the high increase in copper levels and the dark discolouration of the oil had taken place within the relatively short period of fewer than 10,000 operating hours. Additives break down or lose some of their effectiveness, while synthetic base oils also deteriorate. Contaminants or lubricating grease often find their way into the oil as well. Most processes interact with one another.
In addition, the various base oils or additives of individual production batches, other operating temperatures or conditions, altered operating times, or stop-and-go operation can often have their own influence on oil ageing. These alternations are all summarised in the lab report under the heading of „Oil Ageing“. Just as varied as these factors are the traditional parameters of lubricant analysis, most of which yield values through standardised methods. It is with the help of these factors that the oil‘s condition or degree of ageing is assessed. It is only possible to make a reliable diagnosis when the various informational elements acquired are linked together. When it comes to the ageing of gear oils, appearance and colour are assessed subjectively in addition to comparing a change in viscosity and viscosity index, acid content and still-present additives with the values of a fresh oil sample. Oxidation is also revealed within a spectrum made visible using FT-IR (Fourier-transform infrared) spectroscopy.
The proven FT-IR method and its limits
The FT-IR spectrum of a used oil sample provides information about changes to the oil or oil mixtures (albeit mostly only in comparison with the spectrum of a corresponding fresh or reference oil). For example, oxygen bonds that change within a sample can deliver a diagnosis of oil oxidation. Although this standardised process has now become established for classic mineral oils, FT-IR spectroscopy can no longer be used to reliably quantify oxidation for synthetic oils, the base oils of which are based on PAO (poly-alpha olefin) or Discolouration of the synthetic gear oil 12,600 operating hours 43,800 operating hours 52,700 operating hours 7 PAO with an ester base, or the additives of which contain synthetic components like these. Spectrum comparison can only be used to determine oil ageing if the molecules present in the lubricant absorb the infrared light differently at specific wavelengths. In accordance with the applicable standard, the oxidation of a used oil is determined from the differential spectrum of the used and fresh oil at a wave number of 1710 cm-1 and given as a numerical value in A/cm (absorption per cm of oil layer thickness). In the case of a lubricant with a mineral oil-based base oil, a relatively small „peak“ that rises continuously in the used oil spectrum is indicative of increasing oil oxidation. FT-IR spectroscopy, which works on the basis of oxygen concentrations, provides no clear information in the event that the oils or additives include ester-containing components. The ester-containing components conceal an oxidation peak in the FT-IR spectrum due to the fact that an oversized peak forms in an ester-containing oil in a wave number region of around 1740 cm-1. This is why infrared spectra of synthetic fresh oils mostly display an oversized „peak“ in a wave-number region around 1740 cm-1. If the spectra of fresh and used oil are compared with one other, any „peak alteration“ caused by oil oxidation is no longer visible.
When appearances are deceiving
A statement with respect to oxidation provides information about the general ageing condition of the lubricant. In the case of the synthetic gear oil described here, however, a safe diagnosis was difficult to make. Synthetic oils often show conspicuous changes over a period of several thousand operating hours in the form of increasing wear or oil ageing. However, some wear values – especially those for non-ferrous metals such as copper and zinc – increase unexpectedly and significantly without prior warning. This is often accompanied by a dark discolouration of the oil. Even so, this colour change alone is no indication of a critical change in the oil. It is only cause for alarm if the darkened oil displays unexpectedly high copper and zinc values.
Faster, more accurate results with HPLC
A traditional oil analysis provides valuable indications as to changes in the lubricant and its effects, providing the maintenance engineer with timely information about any change in viscosity, rising water content, acids in the oil, or contaminants. However, the question of why the darkened oil in our example showed such a high copper content could not be answered with the analysis included in a traditional OELCHECK analysis kit. Instead, the OELCHECK tribologists had to develop an all-new approach. To inspect the gear oil, they employed high performance liquid chromatography (HPLC) for the first time. The HPLC device is normally used to test whether the expected number of inhibitors is still present within a coolant. An inhibitor is added to a coolant to slow down or inhibit chemical or physical changes.
The inhibitors used are azoles like tolytriazole and/or benzotriazole, which protect components with non-ferrous metal-containing surfaces from corrosion. Non-ferrous metal inhibitors are also used to protect surfaces in lubricants. These additives can, however, break down and lose effectiveness as usage time continues. If it is possible to detect the breakdown of these protective ingredients accurately and ahead of time, an impending corrosive attack on the lubricated elements can be identified and documented. Non-ferrous metal inhibitors cannot be identified with an element analysis, FT-IR spectroscopy or other standardised process. With high performance liquid chromatography, however, OELCHECK already has the perfect laboratory device in place. The two synthetic gear oil samples taken at 43,000 and 52,700 operating hours, respectively, were tested together with a fresh oil sample using the HPLC method. The results confirmed the OELCHECK tribologists‘ hunch. The HPLC graph for the fresh oil (blue) shows its high tolytriazol content. A significant decline is evident in the graph for the sample taken after 43,800 operating hours (grey). Finally, after 52,700 operating hours (red), the content of the non-ferrous metal inhibitor tends towards zero. In other words, the wear has already begun. This information about the significant decline in the non-ferrous metal inhibitor at 43,000 operating hours would have made it possible to halt the rising copper wear in time, e.g. by performing an oil change.
Examples in the OELCHECK database, which contains over 3 million data records, shows that increases in non-ferrous metal content can be even more striking. Sample comparisons show that copper and zinc values have risen within the space of fewer than 2,000 operating hours to concentrations of well over 100 mg/kg without a cause being identified. With the HPLC method, OELCHECK now has a method at its disposal for identifying non-ferrous metal wear at an earlier stage. The process itself is relatively expensive due to the fact that after every analysed sample, the device has to be recalibrated with the oil to be inspected; however, these advance warnings will enable damage and cost-intensive repairs to be avoided.
Non-ferrous metal inhibitor content (NFMIC) available as a special test
A traditional oil analysis accurately determines the oil condition and wear as part of an analysis kit. With the examination of non-ferrous metal inhibitor content, an additional method has been made available with results that enable any corrosive wear procedures at work in non-ferrous metal components to be identified earlier than was previously possible. OELCHECK recommends the new NFMI procedure for the analysis of gear oils in order to improve operating safety, especially in the event that an elevated non-ferrous metal component (copper, lead, tin) is identified. With the analysis of NFMI content using the high performance liquid chromatography (HPLC) method, the remaining residual amount of non-ferrous metal inhibitor is given as a percentage in comparison with the value from a fresh oil sample in the „Additional Tests“ section of the OELCHECK lab report.
OELCHECKER Spring 2019, pages 6-7